A lead-acid battery is the most inexpensive battery and is widely used for commercial purposes. It consists of a number of lead-acid cells connected in series, parallel or series-parallel combination.
A lead-acid cell basically contains two plates immersed in electrolyte (dilute sulphuric acid i.e. H2SO4 of specific gravity about 1.28). The positive plate (anode) is made up of lead-peroxide (PbO2) and the negative plate (cathode) is made up of sponge lead (Pb).
When the cell is delivering electrical energy to the external circuit (load), the process is known as discharging of the cell. Whereas, when it is taking electrical energy from the external DC source, the process is known as charging of the cell.
Charging and Discharging of Lead Acid Battery
Discharging of Lead Acid Battery
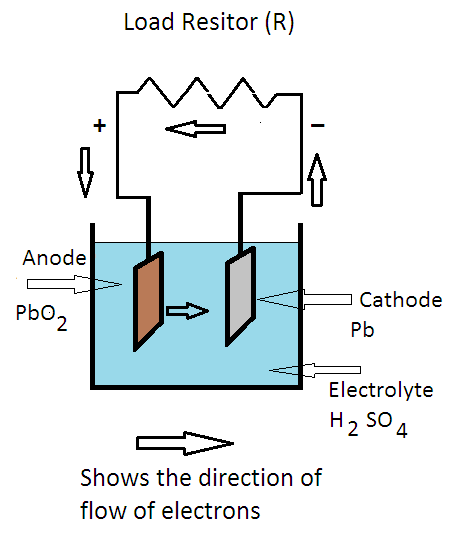
When the sulphuric acid is dissolved, its molecules are dissociated into hydrogen ions (2H+) and sulfate ions (SO4– –) which moves freely in the electrolyte.
When the load resistance is connected to terminals of the battery; the sulfate ions (SO4– –) travel towards the cathode and hydrogen ions (2H+) travel towards the anode. The chemical reactions at the cathode are as under:
At cathode
SO4– – – 2e = SO4 (radical)
Pb + SO4 —–> PbSO4
At anode
Each hydrogen ion (H+) on reaching the anode, takes one electron from it to become hydrogen gas. This electron is the same electron which is given by the sulfate ion at the cathode and has come to the anode via external load resistor R.
2H+ + 2e = 2H
The hydrogen gas liberated at the anode acts chemically on the anode material (PbO2).
PbO2 + 2H —-> PbO + H2O
This PbO2 is reduced to PbO which reacts with H2SO4 and forms PbSO4.
Thus during discharging
- Both the plates are transformed into lead sulfate (PbSO4).
- Sulphuric acid is consumed and water is formed which reduces the specific gravity of electrolyte from 1.28 to 1.18.
- The terminal voltage of each battery cell falls to 1.8V.
- Chemical energy is converted into electrical energy which is delivered to load.
Charging of Lead Acid Battery
The lead-acid battery can be recharged when it is fully discharged. For recharging, positive terminal of DC source is connected to positive terminal of the battery (anode) and negative terminal of DC source is connected to the negative terminal (cathode) of the battery.
During recharging, hydrogen ions (2H+) travel towards the cathode and sulfate ions (SO4– –) travel towards the anode. The chemical reactions are as under:
At anode
SO4– – – 2e —> SO4 (radical)
SO4 + H2O —> H2SO4 + O
The oxygen (in atomic stage) reacts chemically with anode material (PbSO4):
PbSO4 + O + H2O —> PbO2 + H2SO4
At cathode
Each hydrogen ion (H+) on reaching the cathode, takes one electron from it to become hydrogen gas. This electron is given by sulfate ion at the anode and has come to cathode via external circuit (from the DC source)
2H+ + 2e —> 2H
The hydrogen gas liberated at the cathode acts chemically on the cathode material (PbSO4)
PbSO4 + 2H —> Pb + H2SO4
Thus, PbSO4 is transformed to Pb. Water (H2O) is consumed and H2SO4 is formed in above chemical reactions. This increases the specific gravity of the electrolyte. Ultimately, the cell regains its previous (before discharging) chemical compositions.
Thus during charging
- The anode is transformed into lead peroxide (PbO2) and cathode into the spongy lead (Pb).
- Water is consumed and sulphuric acid is formed which increases the specific gravity of electrolyte from 1.18 to 1.28.
- The terminal voltage of each battery cell increases to 2.2 to 2.5V.
- Electrical energy is converted into chemical energy which is stored in the cell.
Lead-acid batteries are charged by:
- Constant current method, and
- Constant voltage method.
In the constant current method, a fixed value of current in amperes is passed through the battery till it is fully charged.
In the constant voltage charging method, charging voltage is kept constant throughout the charging process. The charging current is high in the beginning when a battery is in a discharged condition, and it gradually drops off as the battery picks up charge. While charging a lead-acid battery, the following points may be kept in mind:
- The source, by which battery is to be charged must be a DC source.
- The positive terminal of the battery charger is connected to the positive terminal of battery and negative to negative.
- The charging current should be set at proper value preferably at the value recommended by the manufacturer. If it is not known then the charging rate should be such that full charge can be obtained in 8 hours. Thus an 80 AH battery should be charged at the rate of 80/8= 10 A. This will ensure the maximum life of the battery. If the charging current is too high, it may be harmful to the battery.
- The temperature of batteries should be watched carefully during charging and not allowed to exceed 43oC – 45o The charging current should be reduced if the temperature exceeds this limit. A high temperature is harmful to separaters and active material of plates.
Charge Indications While Lead Acid Battery Charging
While lead acid battery charging, it is essential that the battery is taken out from charging circuit, as soon as it is fully charged. The following are the indications which show whether the given lead-acid battery is fully charged or not.
- Voltage : During charging, the terminal voltage of a lead-acid cell When the terminal voltage of lead-acid battery rises to 2.5 V per cell, the battery is considered to be fully charged.
- Specific gravity of Electrolyte: When the cell is fully charged, specific gravity of electrolyte rises to 1.28 which can be measured with the help of a hydrometer.
- Gassing: When the cell is fully charged, the charging current starts electrolysis of water. The result is that hydrogen and oxygen gases are liberated at the cathode and anode respectively. This liberation of gasses at both the plates is known as gassing. The bubbles thus formed indicate that the cell is fully charged.
Lead Acid Battery Safety Precautions
- If the level of electrolyte decreases, add distilled water. Addition of electrolyte will be necessary only if some of the electrolyte leaks or spils out. The level of the electrolyte should always be above the plates by at least one-half inch.
- Do not over charge a battery.
- Do not deep discharge a battery.
- The gases, hydrogen and oxygen, issuing from a battery under charge can explode if a spark or flame is brought too near.
- The batteries should be charged in a well-ventilated place so that gases and acid fumes are blown away.
- The lead-acid battery should never be left idle for a long time in discharged condition because the lead sulfate coating on both the positive and negative plates will form into hard crystals that will be difficult to break up on recharging. Although it can be left idle for some time in charged condition.
- If acid falls on a piece of cloth, it should be washed with water first and then with liquid ammonia.
Thanks for reading about “charging and discharging of lead acid battery”.